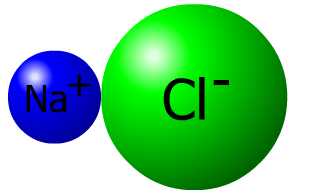
Ionic Bonds
|
This interactive activity from ChemThink discusses ionic bonding—a type of chemical bond formed between two ions with opposite charges. Investigate how the transfer of electrons between atoms creates ions and how the mutual attraction of these charged particles forms ionic bonds. Also learn about trends in the periodic table of elements, and explore how the structure of an ionic compound relates to its formula.
|
- Ionic compounds contain ionic bonds formed by the attraction of oppositely charged ions
- Ions in an ionic compound are arranged in a repeating pattern known as a crystal lattice
- Ionic compound properties are related to ionic bond strength
- Polyatomic ions consist of more than one atom and act as a single unit
- Ionic compounds are electrolytes; they conduct an electric current in the liquid phase and in aqueous solution