Metallic Bonds
|
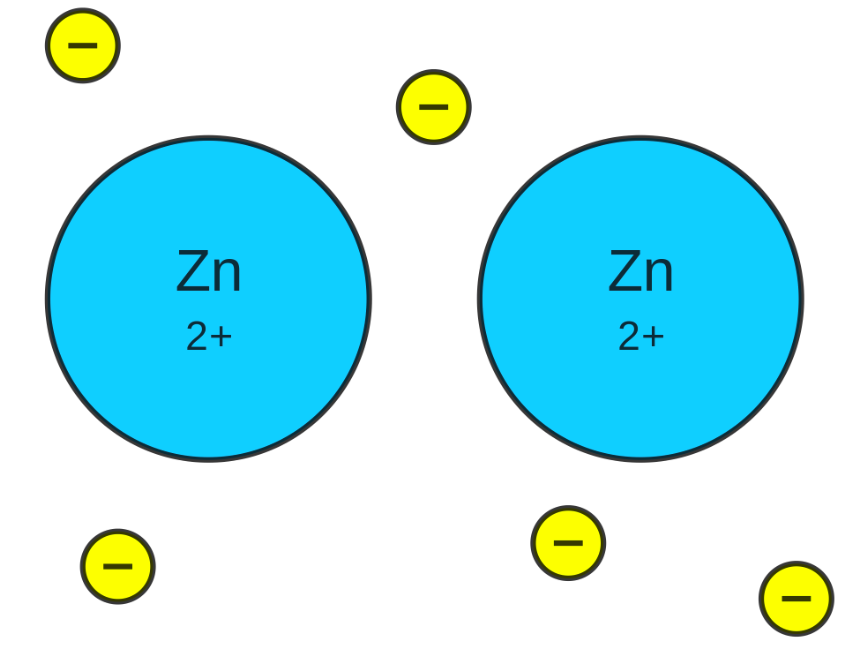
In a metallic bond, one or more of the outer electrons of the atoms are "delocalized." In other words, the electrons are free to move around all the atoms involved, surrounding the atoms with a cloud of valence electrons. The strong attraction between the electron cloud and the positively charged atoms binds the atoms together, creating a metallic bond.
|
- A metallic bond forms when metal cations attract freely moving, delocalized valence electrons
- In the electron sea model, electrons move through the metallic crystal and are not held by any particular atom
- The electron sea model explains the physical properties of metallic solids
- Metal alloys are formed when a metal is mixed with one or more other elements